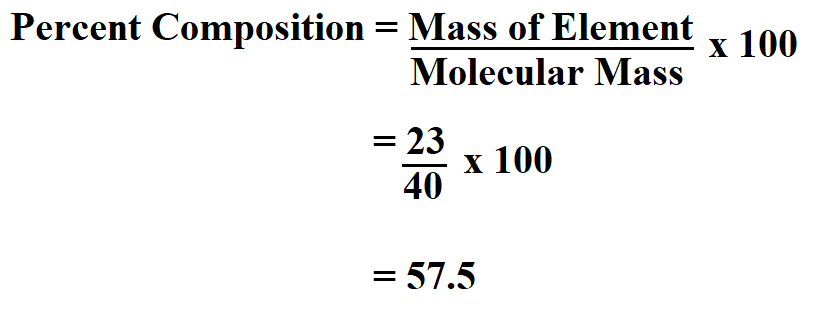
Choose a mathematical equation to find the unknown Extract the information from the question To answer each question correctly you will need to: In each of the worked examples below, you will be asked to calculate either the moles, mass, or molar mass of a pure substance. Worked Examples of Calculating Mass, Moles, Molar Mass Substitute in the values and solve the equation to find the molar mass of the substance in grams per mole. Therefore, molar mass (g mol -1) = mass (g) ÷ moles (mol)įollow these steps to calculate the molar mass of a pure substance given the amount of substance in moles and the mass of substance: Substitute in the values and solve the equation to find moles of substance (mol).īy inspection of units we see that dividing the mass in grams by the amount in moles we arrive at a quantity with the units grams per mole (g mol -1) which are the units for molar mass. If mass is given in kilograms (kg), multiply it by 1,000 to give a mass in grams (g). If mass is given in micrograms (μg), divide it by 1,000,000 to give a mass in grams (g).
If mass is given in milligrams (mg), divide it by 1,000 to give the mass in grams (g). Mass = m = write down what you are told in the question If we turn this upside down (in mathematical terms, take the reciprocal) we get a quantity with units of "mol" which is what we want:įollow these steps to calculate the amount of pure substance in moles given the mass of substance: Substitute in the values and solve the equation to find the value of mass, m, in grams (g).īy inspection of units we see that dividing molar mass by mass will give us a quantitiy in units of "mol -1" Write the mathematical equation (mathematical formula): If amount is given in kilomoles (kmol), multiply it by 1,000 to give an amount in moles (mol). If amount is given in micromoles (μmol), divide it by 1,000,000 to give an amount in moles (mol). If amount is given in millimoles (mmol), divide it by 1,000 to give the amount in moles (mol). The amount of substance must be in moles (mol) ! Check the units for consistency and convert if necessary: (you may need to calculate this using the molecular formula of the pure substance and a Periodic Table) Molar mass = M = write down what you are told in the question (units are g mol -1) Moles = n = write down what you are told in the question Mass (g) = molar mass (g mol -1) × moles (mol)įrom the data in the table and its graphical representation, we can generalise and say that for any pure substance the mass of substance in grams is equal to the moles of substance multiplied by the mass of 1 mole of the substance:Īnd since mass of 1 mole of a substance (in grams) = molar mass (in grams per mole)įollow these steps to calculate the mass of a pure substance given the amount of substance in moles: Mass (H 2O) = molar mass (H 2O) × moles (H 2O) Since 18.016 g mol -1 is the molar mass of water, we can say: We can determine the slope of the line using 2 points on the straight line, for example, (0,0) and (15.0, 270.24): Slope (gradient) of the line = vertical rise ÷ horizontal run This graph above shows a straight line that passes through the origin (0,0) so the equation for the line is: The table below compares the mass of different amounts of water in moles:

Mass water = moles of water × molar mass of water Mass water = moles of water × mass of 1 mole waterīecause the mass of 1 mole of water in grams is known as its molar mass, we can write: In both of the examples above, we can calculate the mass of water in grams by multiplying the moles of water by the mass of 1 mole of water in grams: Mass of ½ mole of water = ½ × mass of 1 mole of water If 1 mole of water has a mass of 18.016 g, then ½ mole of water must have ½ the mass: So, if we only had ½ mole of water, what mass of water would we have? Mass of 10 moles of water = 10 × 18.016 = 180.16 g (about the mass of water you could put in a small glass) Mass of 10 moles of water = 10 × mass of 1 mole of water If 1 mole of water has a mass of 18.016 g, then 10 moles of water must have ten times more mass: From the table we see that 1 mole of water has a mass of 18.016 grams, which isn't very much (about the mass of water in a couple of small ice-cubes you'd make in your family freezer).īut what if you had 10 moles of water? What would be the mass of 10 moles of water?
